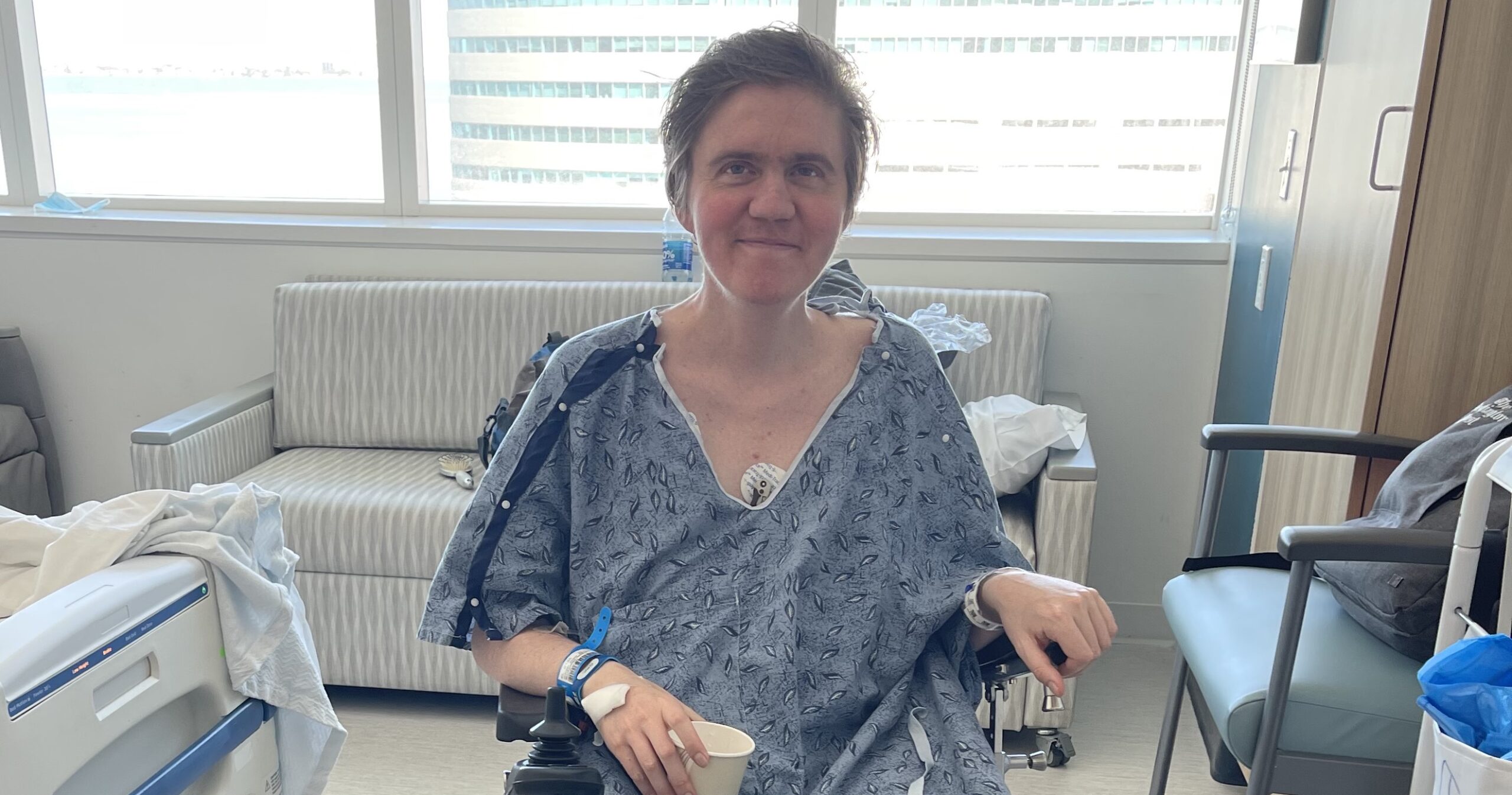
Hawken Miller, who brings his journalism experience and the perspective of having Duchenne himself, writes about how he tries to focus on the things that he can do rather than what he can’t in this CureDuchenne blog. It helps him live a more purposeful and joyful life.
The last day of FUTURES 2023 was jam-packed with sessions focused on how people with Duchenne can use their lived experience to help others going through the same challenges. A Game of Life encouraged people to share tips and tricks that work for them, breakout sessions met people wherever they were at in their Duchenne journey, and the closing session was an opportunity for everyone to share what was on their mind.
Today was all about ensuring that boys and adults with Duchenne receive the best care possible. It was also a time for celebration. At the end of the day, CureDuchenne hosted a summer camp-themed party for individuals with Duchenne, parents, caregivers, and anyone tied to the cause.
The first full day of FUTURES has wrapped! In the last couple of hours, we’ve had educational workshops on gene therapy, RNA-based therapeutics, and muscle restoration and anti-inflammatory treatments. Plus, everyone’s had a chance to meet a new person and create relationships that will last a lifetime. It’s been a lot to digest, so here’s the top highlights from Friday at CureDuchenne FUTURES.
San Diego is one of the most accessible cities in the world, with a fully accessible trolley system laid out along a grid. But there’s still some things that are […]
There’s plenty of times in life where plans fall apart. That’s especially the case if you have Duchenne muscular dystrophy like me. A building you were expecting to enter might […]
Hawken Miller is a journalist, bringing his experience writing for outlets like The Washington Post and BioNews, along with his personal experience living with Duchenne muscular dystrophy, to CureDuchenne. Learn more about Hawken here.
Find a clinical trial that works for you! Clinicaltrials.gov, managed by the U.S. National Library of Medicine, includes 439,599 research studies in 50 states and 221 countries. Many of those […]
Attending a concert or sporting event is a fun activity for everyone. With proper planning and research, people with Duchenne can also participate and enjoy their favorite band or sports team with their friends.