WMS 2021 Day 4: Clinical Trial Updates

WMS 2021 on Thursday, September 23rd was a full day of oral and poster presentations, and we’ll highlight some Duchenne clinical trial updates covered.
Solid Biosciences – SGT-001 micro-dystrophin gene therapy
Solid Biosciences reported 1.5-year follow-up data for three individuals (aged 6-11 years at the time of treatment) receiving high-dose (2xE14vg/kg) SGT-001 in the ongoing Phase I/II clinical trial.
In May of this year, Solid presented biopsy data from these individuals (taken 1-2 years post-dosing) showing widespread and durable expression of the micro-dystrophin protein.
Newly presented today was encouraging functional data and patient-reported outcomes at 1.5 years:
- NSAA scores showed minimal change compared with baseline
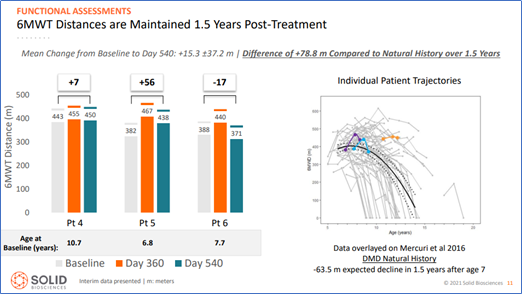
- Six-Minute Walk test distances were maintained
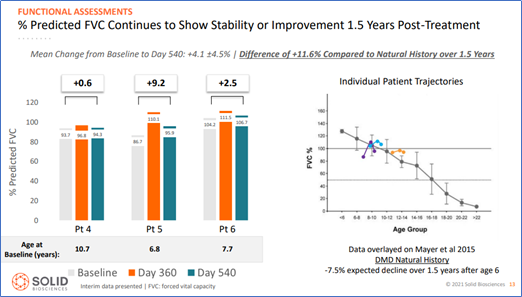
- Respiratory FVC measure continued to show stability or improvement
Given that natural history analyses indicate that declines in function would typically be observed during the same time period, these results suggest functional benefits after treatment with SGT-001.
Patient-reported outcomes as measured by the PODCI (Pediatric Outcomes Data Collection Instrument) also showed sustained improvements compared to baseline, while natural history data indicates individuals would typically demonstrate declines.
In all, these data are encouraging in that SGT-001 may confer meaningful improvements to individuals with Duchenne. Two more participants have already been treated with the same dose but have not yet reached the 1-year follow-up timepoint.
ReveraGen Biopharma – Vamorolone
In the “late-breaking news” section, Guglieri et al. reported results from a 24-week double-blind randomized study examining vamorolone versus placebo and prednisone treatment in Duchenne patients aged 4 to <7 years old.
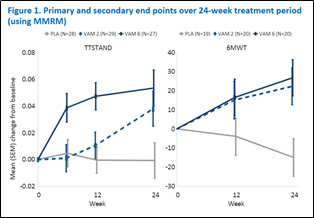
The primary efficacy outcome measure was changed from baseline to week 24 for Time to Stand (TTSTAND) velocity, while secondary outcome measures included change in baseline Six-Minute Walk test (6MWT), NSAA, myometry, and others.
The study met its primary endpoint TTSTAND velocity (6 mg/kg/day vamorolone vs placebo, p = 0.002), in addition to the first four sequential secondary endpoints.
Of note, the prednisone group, but not the vamorolone groups showed linear growth delay (vamorolone 6 mg/kg/day versus prednisone p=0.02), and vamorolone-treated groups showed no significant decreases from baseline on bone remodeling biomarkers at 24-weeks treatment.
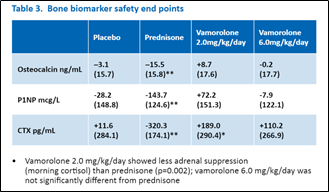
These late-breaking results demonstrate somewhat comparable efficacy between vamorolone and prednisone, but meaningful differences with vamorolone treated boys showing normal growth trajectories and no reduction in biomarkers of bone remodeling. Longer-term studies will evaluate fracture rates between vamorolone and corticosteroid treatment.
These findings are consistent with results from earlier studies from ReveraGen, which are currently in long-term extension. In the Clinical Trials oral session at WMS 2021, ReveraGen presented results from an open-label long-term extension study, including 2.5 years of vamorolone treatment in 46 individuals.
Since some individuals started at low doses of vamorolone for the first 6 months, before switching to higher doses, a delayed start analysis was employed. As shown below, there are two groups for each measure – “High dose” treated with 2 or 6 mg/kg/day for the full 2.5 years, or “Low dose” who switch to high dose at month 6 and have 18-23 months of high dose treatment. The functional measures all show inflection points at the transition from low dose to high dose, providing additional support for a treatment effect.
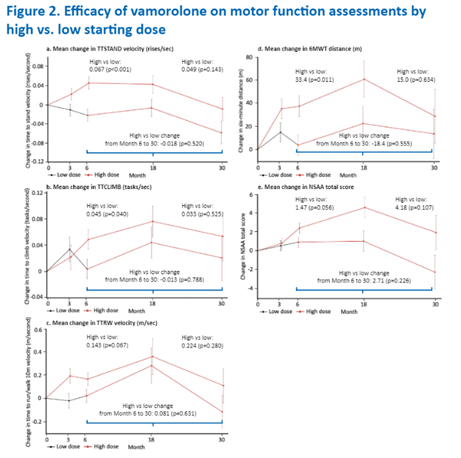
The modest functional improvements in TTCLIMB velocity, TTRW, NSAA, and 6MWT over the 2.5 years, and the improvement to 18 months then a decrease in TTSTAND velocity is consistent with what is seen in boys with DMD treated with glucocorticoids.




