What is Duchenne?
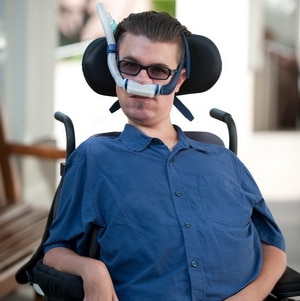
Duchenne is a devastating muscle disease in children. Historically, most boys who have it do not survive beyond their late twenties, and those that do will be using a wheelchair by age 12 and experience social isolation. The simplest of tasks become difficult, and in the later stages, heart and breathing muscles begin to fail. Nearly 20,000 boys are living with the disease in the United States alone and over 300,000 worldwide. The disorder knows no cultural, economic, or social boundaries.
Muscular dystrophies are a group of genetic disorders characterized by muscle weakness. Duchenne is the most common and severe form, caused by loss of dystrophin, beneath the sarcolemna. The molecular mechanisms of the disease have been extensively investigated since the discovery of the gene in 1986.
The good news is that new gene-based therapies have recently emerged with noted advances in using conventional gene replacement strategies, RNA-based technology, and pharmacological approaches. In particular, antisense-mediated exon skipping has shown encouraging results and holds promise for the treatment of dystrophic muscle.
Who We Are
We are committed to improving the lives of everyone affected by Duchenne through accelerating research to find the cure, improving care and empowering the Duchenne community.
CureDuchenne is recognized as the global leader in research, patient care and innovation for improving and extending the lives of those with Duchenne. We operate with integrity through compassion, transparency and empowering the community nationally with positive solutions.
Our actions fuel hope for families, enable progress for drug development, and extend ambulation for patients in collaboration with pharmaceutical companies, medical and healthcare professionals, our scientific advisory board, and our board of directors.
Research Funding
The funds we raise support the most promising research programs aimed at treating and curing the disease. We have also garnered international attention for our efforts to raise funds and awareness through venture philanthropy (CureDuchenne Ventures). For the first time, there is hope for this destructive disease affecting thousands of children and their families.
We’ve leveraged $100 million in pharma and biotech research and development investments. To date, nine CureDuchenne research projects have advanced into human clinical trials. That’s a unique accomplishment. And we contributed funding to Sarepta Therapeutics, the first company to have an FDA-approved drug for Duchenne.
Community Support
Under our CureDuchenne Cares banner, we strive to increase the health and wellbeing of patients living with Duchenne through educating parents, caregivers, clinicians and physical therapists on best practices. Our goals are to improve outcomes, extend ambulation, and offer guidance on disease management and care.
This program also keeps families informed on the latest scientific developments in Duchenne research, including status and participation in clinical trials. We provide workshops and webinars on a wide range of topics. And we’ve created the CureDuchenne Cares Certified Physical Therapist program to ensure high standards of care. In short, we strive to build a community where all parties involved can share information, get the resources they need and find mutual support.
