WMS 2021 Day 1: Industry Symposia

Monday, September 20th was the first day of the WMS 2021 Virtual Congress, and a day devoted to symposia sponsored by various companies developing therapeutic treatments for neuromuscular disorders. Most relevant to Duchenne was the symposia sponsored by PTC Therapeutics and by Sarepta Therapeutics.
PTC’s Symposia
PTC developed Translarna (ataluren) to address nonsense mutations in the gene causing Duchenne muscular dystrophy. A nonsense mutation is an alteration in the genetic code that causes formation of the intended protein, dystrophin, to be prematurely stopped, and PTC’s drug aims to enable this stop signal to be ignored. Although it is approved for use in DMD in various countries including in the UK and Europe, Translarna is not approved for use in the United States.
As part of an effort to gather more data on the safety and efficacy of Translarna, the STRIDE registry has been collecting real-world evidence across 13 countries in Europe and Israel. Today, PTC presented 5-year data from 241 boys treated with Translarna and showed that treatment delayed the loss of ambulation by more than 5 years, as compared to data from a similar group of individuals in the CINRG natural history study. The median age of loss of ambulation was 17.9 years for those treated with Translarna, compared to 12.5 years old for individuals not treated. At 12 years old, 80% of boys receiving Translarna were walking, compared to 52% of the boys in the untreated, comparator group.
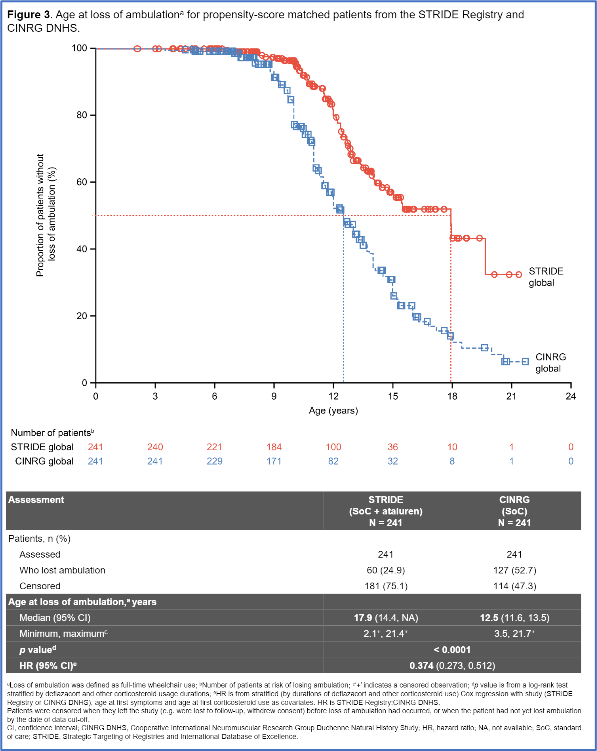
Delays in loss of respiratory function were also observed. The age at which individuals crossed a critical respiratory threshold often signaling need for respiratory physical therapy was 17.6 years old in the treated group, compared to 15.8 years in those not receiving Translarna.
PTC issued a press release also announcing these findings. PTC has 5 posters at WMS 2021, and show that Translarna treatment delays decline in ambulation, some North Star Ambulatory Assessment (NSAA) items and respiratory measures.
Sarepta Symposia
Sarepta provided an overview of all of its neuromuscular disease programs, including both exon-skipping and gene therapy approaches in Duchenne muscular dystrophy. In total, Sarepta has nine poster presentations at WMS, but one of the most notable was teased in today’s symposia, and was an update on their gene therapy clinical trial in Duchenne.
Sarepta provided their long-awaited 3-year safety and functional outcome update from those patients (4 to 7-years old at the initiation of the trial) who participated in their Phase 1/2a gene therapy study of SRP-9001 (rAAVrh74.MHCK7.mdystrophin).
The data demonstrated that SRP-9001 was well tolerated over the 3-year period, with no new safety concerns noted after the first 90-days post-infusion.
Importantly, long-term functional assessment exhibited a durable response, as measured by NSAA, showing an overall clinically meaningful improvement in motor abilities in each patient over the 3-year period compared to their baseline values.
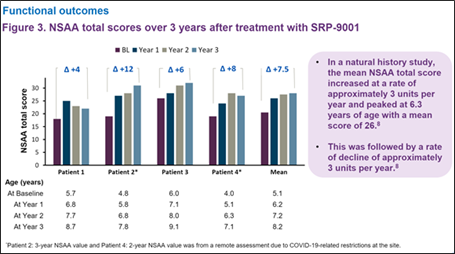
Additionally, patients generally maintained muscle strength as measured by Time to Rise and 4-Stair Climb, and generally improved ambulation (100m walk test).
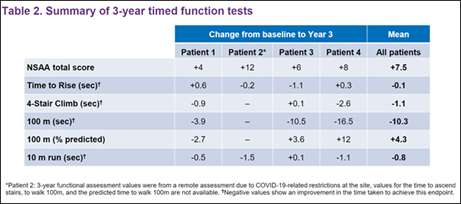
These results demonstrate a safe and durable response to rAAVrh74.MHCK7.mdystrophin gene therapy in Duchenne patients over the first 3-years following treatment.
Worldwide Phase 3 registration trials are now in progress.




